WingRight
adult stem cells
Generation of blood vessel with patient’s own stem cells.
 In the article, “To Fix a Heart, Doctors Train Girl’s Body to Grow New Part,” the Wall Street Journal reported yesterday on an adult stem cell treatment which may revolutionize care of as many as 3,000 children a year. The story focuses on Angela, now 4 years old, who was born with “hypoplastic left heart syndrome,” a condition in which a child never develops a normal 4 chamber heart.
In the article, “To Fix a Heart, Doctors Train Girl’s Body to Grow New Part,” the Wall Street Journal reported yesterday on an adult stem cell treatment which may revolutionize care of as many as 3,000 children a year. The story focuses on Angela, now 4 years old, who was born with “hypoplastic left heart syndrome,” a condition in which a child never develops a normal 4 chamber heart.
Think of the heart as two tubes, each with an atrium or upper chamber and a lower chamber, the ventricle. The lungs are between the two, and the system is set up so that low oxygen blood does not mix with the blood that is saturated with oxygen. 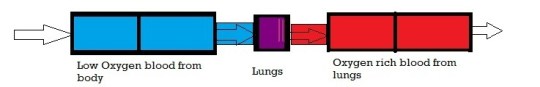
If there is only one ventricle, the system can’t adapt to increased need for oxygen. Not only is the ability to pump the blood not enough to increase when exercising, excited, or scared – or even growing – but the oxygen-rich blood always mixes with the depleted blood, even on the way to the lungs.
Without surgery, 70% of children die before they turn one year old. Up until now, the corrective surgery has involved using grafts of artificial tubes, that don’t grow, and need to be replaced every so often.
The new technique uses a “bio-absorbable” scaffold on which the patient’s own stem cells are seeded and grown. This new blood vessel is surgically implanted so that the anatomy, and the blood flow, is more like the normal heart. It’s hoped that the new graft will grow with the child and prevent the need for repeat surgeries as the child grows.
In the end, the choice to become the first patient in Dr. Breuer’s study turned on three things, Ms. Irizarry says: the family’s faith in God, their trust in the doctor, and the potential for a natural blood vessel that could possibly help avoid more surgeries. “Before, they were using plastic, now they’re using this special graft that will grow with her,” Ms. Irizarry says.
Angela is the first, so we don’t really know whether the new vessel will grow with her. But there’s evidence from other, similar procedures to build new bladders and other organs.
Unfortunately, the process of getting the new procedures from early trials to use for those 3,000 babies a year is complicated and slow, even with an FDA “exemption:”
Development of the procedure has been painstaking. Dr. Breuer undertook four years of laboratory research after he joined Yale in 2003 before seeking approval from the U.S. Food and Drug Administration in 2007 to test the approach on patients. It took four more years and 3,000 pages of data before he got a greenlight. The study builds on the cases of 25 children and young adults successfully treated in Japan a decade ago with a similar approach.
Dr. Breuer, who holds several patents through Yale related to the technology, expects to implant a tissue-engineered blood vessel in a second patient soon as part of a six-patient study to test the safety of the procedure and determine whether the blood vessels actually grow in the body as a child gets bigger. The hope is that if these patients are treated without a hitch, the procedure may be available under a special FDA humanitarian device exemption, which would allow Yale to charge for it while conducting a larger study.
It’s a shame that it took so long to get the procedure to the first patient, and that many more will have to wait even longer. There must be a way to place judgement before bureaucracy in these cases.
Cross posted at LifeEthics.org
Legislator says stem cells helped » Times Record News
This is a wonderful story. I’m very glad for the Representative and for all the patients who receive their own stem cells and have good results. (My granddaughter, at 15 months old in 2001, received an anonymous little boy’s umbilical cord blood after her bone marrow completely failed. More here.)
Someday, I believe we’ll find the stimulating factors that make the body’s stem cells activate the way we want them. In the meantime, this is what our researchers – and Legislators – are finding out about ethical adult stem cells (not destructive embryonic stem cells.:
State Rep. Rick Hardcastle, R-Vernon, participated in a recent round of autologous adult stem cell treatments to help his multiple sclerosis, similar to what Gov. Rick Perry had done in July.
Although the stem cells are not embryonic, doctors in the U.S. are still skeptical of the procedure because it is not yet approved by the Food and Drug Administration.
Adult stem cells are taken from the patient’s fat, sent to a lab where they are developed, then reintroduced to the patient via intravenous therapy.
The treatments are used to treat patients with autoimmune diseases such as multiple sclerosis, fibromyalgia, Crohn’s disease, Parkinson’s and chronic obstructive pulmonary disease.
Hardcastle was diagnosed with MS almost 10 years ago and repeatedly said the treatments worked phenomenally for him.
“I’m walking on water and near bulletproof,” Hardcastle said from a casino in Las Vegas, where he was with his wife for the National Finals Rodeo. “Since I had the third treatment, I have fished in the river in Alaska. I have walked up and down stairs without having to hold onto the handrail like a goon. It’s just been phenomenal so far.”
Hardcastle said just having his balance is an amazing thing because since he was diagnosed, his balance was one of the first things to go. He spoke at length about how easily he was able to walk the stairs at the Las Vegas event.
“Eight years ago, I was having to literally … stop to step over a concrete barrier on a parking curb. I just walk across it now like I did 20 years ago,” he said.
Cloning pioneer urges shift away from embryonic stem cells
Universal Truth at work again. I would have loved to be there in order to watch heads explode and hear the susurus of “Did he say that?” buzzing around the room.
Newer and safer forms of stem cell therapy will likely overtake research into the use of human embryonic stem cells, the scientist whose team cloned Dolly the sheep told his peers at a stem cell conference in La Jolla.
Direct “reprogramming” of adult cells into the type needed for therapy is gradually becoming a reality, Ian Wilmut told an audience of several hundred at the Salk Institute at the annual Stem Cell Meeting on the Mesa. Such a feat was once thought impossible, but in recent years it has been demonstrated in at least two publications, he said.
********
But it’s been unclear which types of stem cells would prove most useful: the “adult” kind that have a more limited potential to change, or the embryonic kind. The emergence of direct reprogramming provides a promising new option scientists should consider, Wilmut said.
“I’m not quite sure why this hasn’t been pursued more actively,” Wilmut said.
It is difficult to achieve purity in embryonic and induced pluripotent stem cells because they are prone to forming tumors.
Direct reprogramming of cells from one type to the other avoids that danger, because the cells never enter the pluripotent stage to begin with, Wilmut said.
Direct cell reprogramming didn’t exist when California voters approved the stem cell program in 2004 with the passage of Proposition 71. That program was mainly aimed at funding embryonic stem cell research the federal government wouldn’t fund.
However, the program can also fund research with other types of stem cells, such as “adult” cells from umbilical cord blood.
The use and value of embryonic stem cells is an intensely controversial issue.
Many people object to their use because human embryos, which they consider human individuals, are killed to get the cells. Critics also point to the success of adult cells in approved therapies, while no therapy with embryonic stem cells has yet been approved.
Only one treatment with embryonic stem cells is in clinical testing in people. And that company, Geron Corp., recently ended its involvement in what was described as a business decision.
via Cloning pioneer urges shift away from embryonic stem cells.
More on the Geron decision to end embryonic stem cell research, here, and on the “Stimulus” funds awarded to Geron and the employees that lost their jobs, anyway, here.
